Downstream System Innovation:
What Is It and Why does It Matter?
Status: Active
Sustainable healthcare systems should reimburse for outcomes rather than products or services. Successfully evolving to such a value-based system in the US requires that we (1) improve our collective ability to get the right treatments to the right patients at the right time, and (2) do this in ways that are economically sustainable for the system. Achieving this dual set of capabilities further requires that we address several daunting challenges in the post-market phase of biomedical innovation:
Knowledge gaps: At the time of regulatory approval of drug therapies, there remain many important uncertainties about drug therapies that can only be progressively reduced by learning from real world use. However, it remains the case that systematic learning about these products stops at the point of regulatory approval. And, where real-world evidence (RWE) is generated, its production is fragmented, costly, inefficient, and not fit-for-purpose for informing key decisions.
Payment for volume, not value: Value-based agreements for drug therapies are a critical component of value-based care, but their effective design and implementation is dependent upon improved outcomes tracking capabilities, as well as, wherever possible, an evidence-based understanding of sub-populations of responders/non-responders.
Fragmentation Across Silos: The above post-market activities (i.e., RWE production and coverage/reimbursement decisions) occur in fragmented ways across operational silos disconnected from each other and from clinical decisions made at the point of care.
These challenges are, to some extent, interdependent. Accelerating the evolution of value-based payment for drug therapies requires the routine availability of high-quality RWE, and enhanced capacity for its efficient and scalable production. But capacity-building in RWE will only be possible when incentives are aligned and all stakeholders benefit. Furthermore, advancements in RWE and value-based payment models must evolve in coordination, with operational linkages that bridge across silos.
All key stakeholders are negatively impacted by these system constraints (Fig 1). Evidence gaps lead to delays in getting the best treatments to each patient, reimbursement for treatments that are less than optimally effective, and commercial risks for developers associated with an undifferentiated marketplace for products in their pipeline.
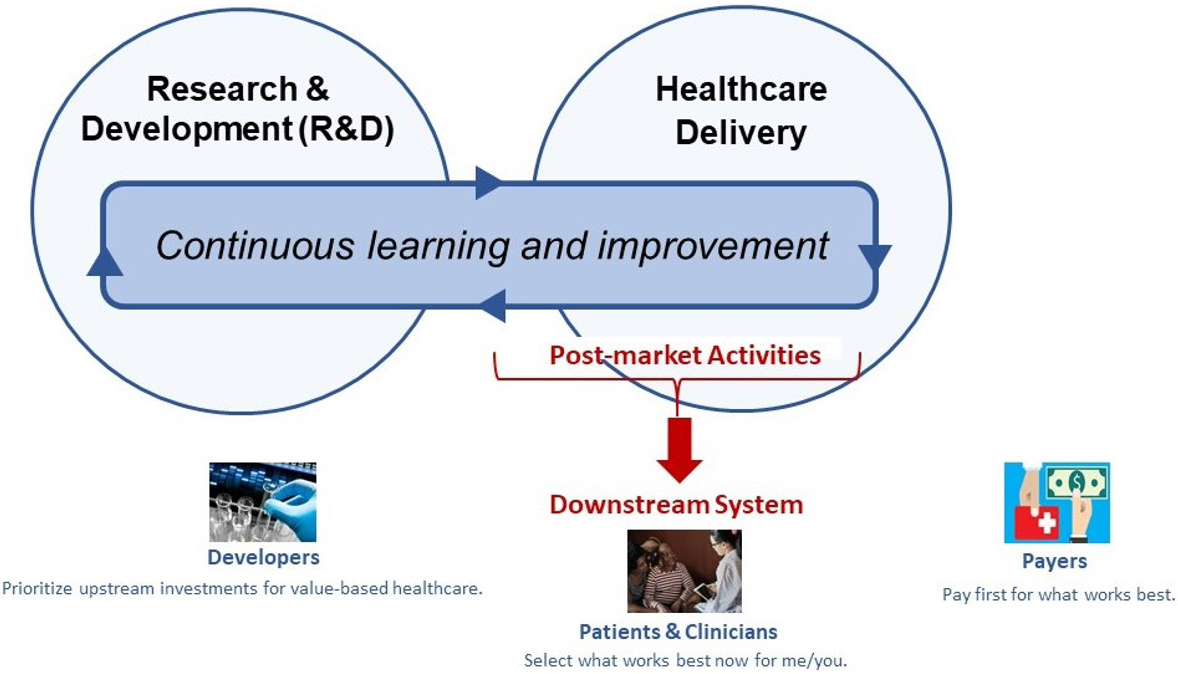
Fig 1: Vision for a “learning health system” as conceptualized by collaborators within NEWDIGS. A core capability includes continuous learning and improvement that spans biomedical R&D and healthcare delivery. Work in NEWDIGS centers specifically on learning that improves decisions and outcomes for all key stakeholders (patients, clinicians, payers, and product developers), and enhancing the operational efficiencies of associated post-market activities through “downstream” system design.
Much of our work in the NEWDIGS consortium has focused on using a case-based approach to explore these challenges associated with the post-market components of the biomedical innovation value chain, specifically RWE production, targeted treatment decisions, and payment for drug therapies.
What is clear from our work is that these activities currently are executed in fragmented, siloed ways, which leads to significant waste and inefficiency. A key goal of our work is to create a downstream (post-market) system where one does not currently exist. We engage end-users (patients, clinicians, payers, biopharmaceutical companies, and regulators, among others) in collaborative Downstream System Innovation in order to more efficiently connect and coordinate RWE generation, targeted treatment decisions, and reimbursement in ways that are both patient-centered and economically sustainable.
We are inspired by noteworthy advancements in Downstream System Innovation by the hemophilia community that has developed a standardized set of outcomes metrics, and the American Thombosis and Hemostatis Network (ATHN) database that supports real-world learning. And by other leading patient-driven, muti-stakeholder initiatives (e.g., multiple myeloma and cystic fibrosis) focused on empowering knowledge generation from natural histories to randomized-controlled trials to post-market learning.
Since our launch in 2009, we have channeled the collective intelligence of our NEWDIGS multi-stakeholder community to advance, for example:
- Payment innovation in the US: Focusing initially on durable cell and gene therapies, we have developed creative payment models that ensure timely and appropriate access for patients; affordability for payers; and sustainability for developers. Tailoring features to address payment timing, product performance uncertainties, and actuarial risks, the models are now being generalized beyond our initial focus, for application to a broad range of diseases and products classes. (More information here).
- How we plan, produce, and use RWE: Getting the right treatment to the right patient at the right time not once, but routinely, will require multi-dimensional innovation in our approach to real-world learning. Our work in “learning health systems” has focused on the design of innovative, platform approaches to real-world discovery of patient sub-populations; adaptive point of care trials for comparative effectiveness; and federated machine learning for predictive models to inform targeted treatment decisions. (More information here).
- Coordinating innovation in payment and RWE: Implementation of payment innovations require new RWE capabilities, and vice versa. NEWDIGS offers the environment, tools, and community to work across traditional silos to coordinate the evolution of payment and RWE models.
Some say that the healthcare system is not actually a system. We believe it is a system - a complex adaptive system (one with many independent players and much uncertainty, and without centralization). Such systems improve through self-organization of players in ways that foster adaptation to emerging information. NEWDIGS excels in developing tools and processes that empower sustainable, patient-centered adaptation of the Downstream System, with a particular focus on connecting RWE, targeted treatments, and payment.